How an ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Physical Properties:
Both Ethanol and ethanoic acid are liquids at room temperature and have distinct melting and boiling points.
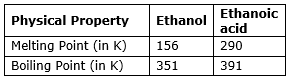
Chemical Properties:
Ethanoic acid produces CO2 when reacted with carbonates or bicarbonates. Ethanol does not react with carbonates or bicarbonates.
Ethanoic acid reacts with a base (say NaOH) to give salt and water:
CH3COOH + NaOH → CH3COONa + H2O
Ethanol reacts very slightly with NaOH.
19