Draw the electron-dot structures of the following compounds and state the type of bonding in each case:
(i) KCl (ii) NH3 (iii) CaO (iv) N2 (v) CaCl2
The dot structure and the bond type of the above given compounds can be shown as
(i) KCl - Ionic bond
![]()
(ii) NH3 - Covalent bond
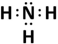
(i) CaO - Ionic bond
![]()
(ii) N2 - Covalent bond
![]()
(iii) CaCl2 - Ionic bond
![]()
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.