State the number of valence electrons, the group number and the period number of each element given in the following table:

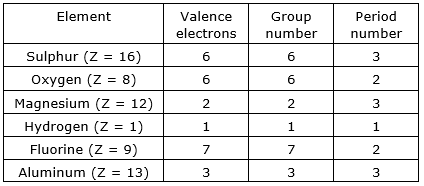
Sulphur
The atomic number of sulphur = 16
The electronic configuration of 16S = 1s2 2s2 2p6 3s2 3p4
⇒ Total number of valence electrons (3s2 3p4) = 6
⇒ As period number is decided by the valence electrons, thus period number of sulphur = 6
⇒ In sulphur, 3 is the highest principal quantum number (n=3), thus group number of sulphur = 3
Oxygen
The atomic number of oxygen = 8
The electronic configuration of 8O = 1s2 2s2 2p4
⇒ Total number of valence electrons (2s2 2p4) = 6
⇒ As period number is decided by the valence electrons, thus period number of oxygen = 6
⇒ In oxygen, 2 is the highest principal quantum number (n=2), thus group number of oxygen = 2
Magnesium
The atomic number of magnesium = 12
The electronic configuration of 12Mg = 1s2 2s2 2p6 3s2
⇒ Total number of valence electrons (3s2) = 2
⇒ As period number is decided by the valence electrons, thus period number of magnesium = 2
⇒ In magnesium, 3 is the highest principal quantum number (n=3), thus group number of magnesium = 3
Hydrogen
The atomic number of hydrogen = 1
The electronic configuration of 1H = 1s1
⇒ Total number of valence electrons (1s1) = 1
⇒ As period number is decided by the valence electrons, thus period number of hydrogen = 1
⇒ In hydrogen, 1 is the highest principal quantum number (n=1), thus group number = 1
Fluorine
The atomic number of fluorine = 9
The electronic configuration of 9F = 1s2 2s2 2p5
⇒ Total number of valence electrons (2s2 2p5) = 7
⇒ As period number is decided by the valence electrons, thus period number of fluorine = 7
⇒ In fluorine, 2 is the highest principal quantum number (n=2), thus group number of fluorine = 2
Aluminum
The atomic number of aluminum = 13
The electronic configuration of 13Al = 1s2 2s2 2p6 3s2 3p1
⇒ Total number of valence electrons (3s2 3p1) = 3
⇒ As period number is decided by the valence electrons, thus period number of aluminum = 3
⇒ In aluminum, 3 is the highest principal quantum number (n=3), thus group number of aluminum = 3
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.

