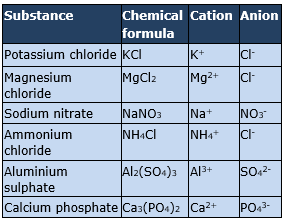
List the cations and anions of the substances given in the table.

Salts are neutral compounds which are ionic in nature and dissociate into positive and negative ions when dissolved in water. The positively charged ion is called cation and the negatively charged ion is called anion. Thus in the above question we need to dissociate the given salts into the respective positive and negative ions, which can be done as follows:
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
