The number of electrons in an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of the element?
Any element is represented symbolically as:
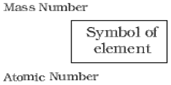
Here mass number = 15 +16 =31
Atomic number = number of protons =15
Hence, the correct representation of the element with 15 electrons and 16 neutrons is ![]() .
.
Hence, the correct option is – A
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.