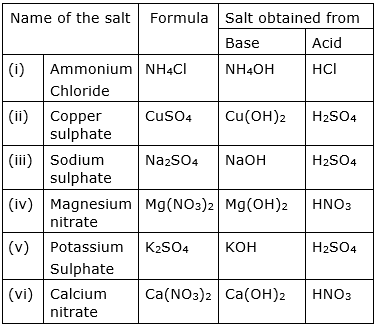
Fill in the missing data in the given table:

Let us write the chemical reactions of each set so as to get the answer for the spaces. These reactions are all neutralization reactions.
(i) 
(ii) 
(iii) 
(iv) 
(v) 
(vi) 
After referring to the above six reactions, the blank spaces in the table can be easily filled.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.