Illustrate an activity to show a double displacement reaction. Give the chemical equation of the reaction that takes place.
Double displacement reaction is a type of reaction which involves the exchange of ions.
Example of Double Displacement Reaction:
Mix ferrous sulphate solution with sodium hydroxide solution. The reaction gives a dirty green precipitate of ferrous hydroxide. The reaction is as follows:
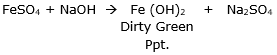
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
