Give chemical tests to detect the presence of a. ethanol and b. ethanoic acid.
(a) Reaction with Na metal:
Ethanol reacts with sodium metal to liberate hydrogen gas.
2R-OH + 2Na → 2R-O-Na+ + H2↑
Iodoform Test:
Ethanol when treated with iodine solution and sodium hydroxide produces a yellow precipitate of iodoform (CHI3).
CH3CH2OH + 4I2 + 6NaOH → CHI3 + HCOONa + 5NaI + 5H2O
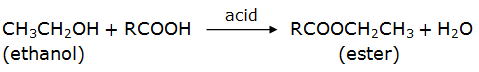
Esterification reaction:
Ethanol reacts with a carboxylic acid in the presence of an acid to produce sweet smelling compounds called esters. The reaction is called esterification reaction.
(b) Reaction with bases:
Ethanoic acid react with bases like NaOH to give salt and water. This is called neutralisation reaction.
Reaction with carbonates and bicarbonates:
Ethanoic acid reacts with metal carbonates and bicarbonates to produce brisk effervescence due to the evolution of carbon dioxide.
2CH3COOH + Na2CO3→ 2CH3COONa + CO2↑ + H2O
CH3COOH + NaHCO3→ CH3COONa + CO2↑ + H2O
Esterification reaction:
Ethanoic acid reacts with an alcohol in the presence of an acid to produce sweet smelling compounds called esters. The reaction is called esterification reaction.
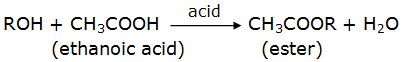
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.