(a) Define the term ‘latent heat of vaporization’ of liquid. What is the value of the of the latent heat of vaporization of water?
(b) Draw a labeled diagram of the experimental set-up to study the latent heat of vaporization of water.
(a) The latent heat of vaporisation of a liquid is the quantity of heat in joules required to convert 1 kilogram of the liquid (at its boiling point) to vapour or gas, without any change in temperature. The latent heat of vaporization of water is 22.5 × 105 joules per kilogram (or 22.5 × 105 j/kg).
(b) 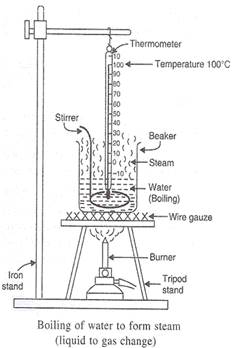
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.