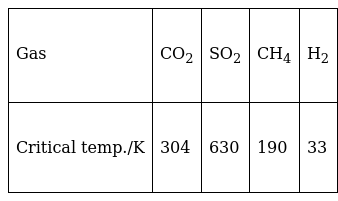
Which of the following is an example of absorption?
Silica gel, finely divided nickel and metal surface all have a porous structure which makes them good adsorbent. Thus they facilitate the adsorption process.
Therefore, (i), (iii) and (iv) are wrong options.
Calcium chloride is a strong hygroscopic substance which absorbs moisture readily. Thus, water is easily absorbed by CaCl2 making it good absorbent.
Therefore, (ii) is correct.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.