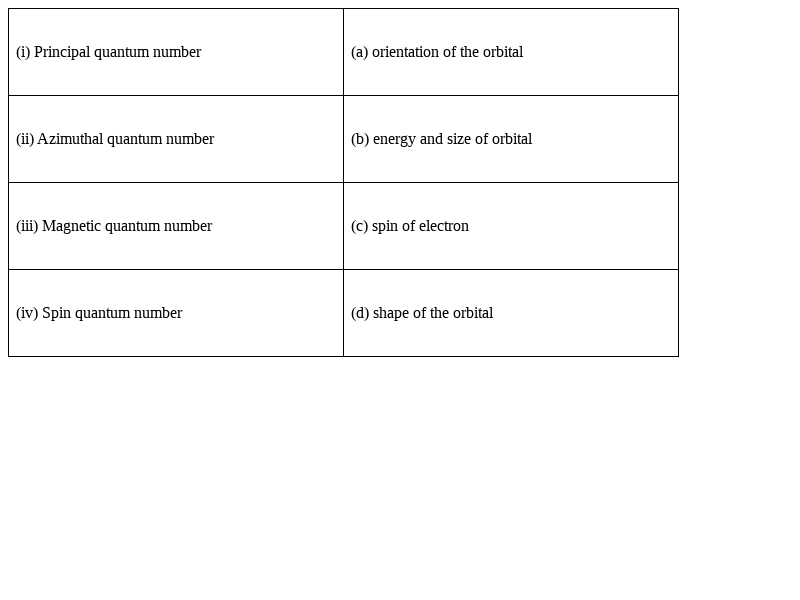
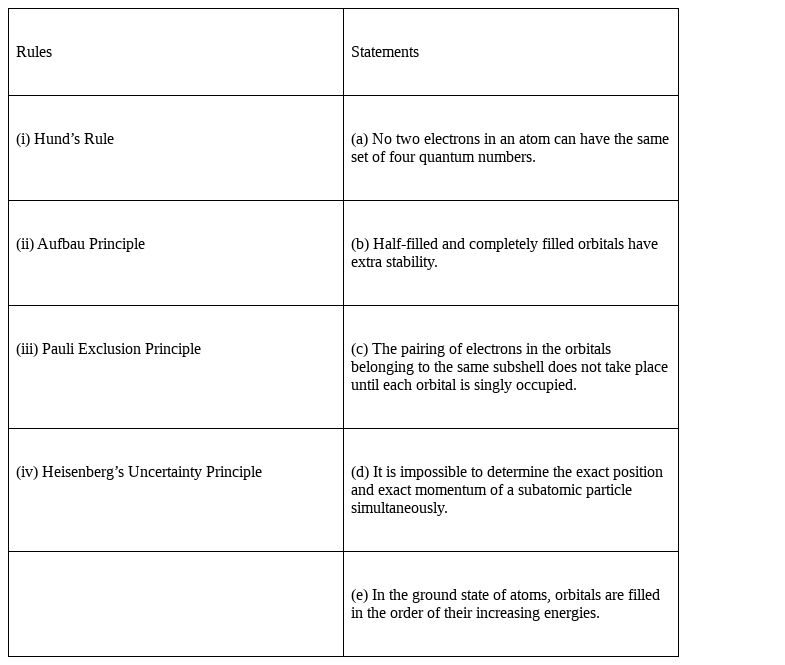
Match the following species with their corresponding ground state electronic configuration.
Atom / Ion Electronic configuration

Matching the following species with their corresponding ground state electronic configuration :
(i)Cu - (c) 1 S2 2s2 2P6 3s2 3p6 3d10 4s1
(ii) Cu2+ -- (d) 1s2 2s2 2p6 3s2 3p6 3d9
(iii)Zn2+ - (a) 1s2 2s2 2p6 3s2 3p6 3d10
(iv)Cr3+ - (e) 1 s2 2s2 2p6 3s2 3p6 3d3
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.