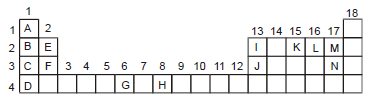
An incomplete form of the periodic table is given below. Write answers to the questions connecting the position of elements in it.

Which is the element with the biggest atom in group 1?
Element D
Atomic size goes on increasing down a group because:
i. Down a group, the atomic number increases one by one.
ii. The outer electrons are adding in a new valence shell.
iii. Therefore, the distance between the outermost electron (valence electron) and the nucleus is also increasing.
iv. Therefore, the size of the atom increases with increase in atomic number.

Number of shells are increasing down a group
Thus, D has the biggest atom in group 1.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.