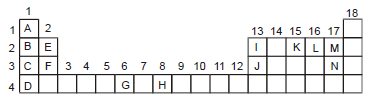
An incomplete form of the periodic table is given below. Write answers to the questions connecting the position of elements in it.

Which of the elements L and M has the lowest electronegativity?
Element L
Both elements L and M belong to the same period.
Electronegativity increases along a period because:
The atomic radius goes on decreasing as we move from left to right due to which the attraction between the outer electrons and nucleus increases.
Also, there is increase in electronegativity with increase in ionization energy. Thus, L has lowest electronegativity.
To remember:
![]()
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.