Give the formulae of the stable binary compounds that would be formed by the combination of following pairs of elements
(a) Mg and N2
(b) Li and O2
(c) Al and Cl2
(d) K and O2
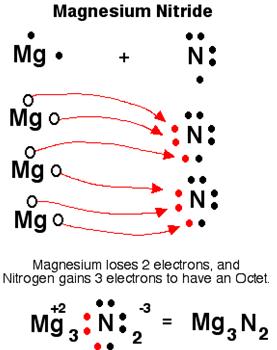
(a) Mg and N2: Mg3N2 (magnesium nitride)
(b) Li and O2: LiO2 (lithium oxide)

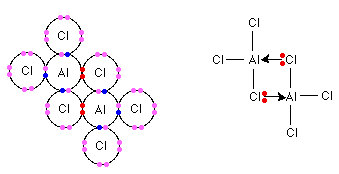
(c) Al and Cl2: Al2Cl3 (Aluminium chloride)
(d) K and O2: K2O (Potassium oxide)

Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.