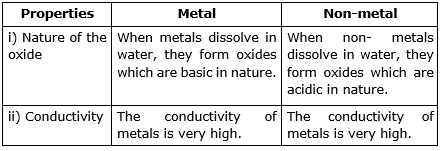
Compare the properties of a typical metal and a non-metal on the basis of the following:
i. Nature of the oxide formed by them
ii. Conductivity
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.