Solution of Chapter 7. Equilibrium (Chemistry - Exemplar Book)
Chapter Exercises
Multiple Choice Questions I
We know that the relationship between Kc and Kp is Kp = Kc (RT)Δn
What would be the value of ∆n for the reaction
NH4 Cl (s) ⇋ NH3 (g) + HCl (g)
view answer >
For the reaction H2(g) + I2(g) ⇋ 2HI (g), the standard free energy is ∆G⊖ > 0. The Equilibrium constant (K ) would be __________.
view answer >Which of the following is not a general characteristic of equilibria involving physical processes?
view answer >PCl5, PCl3 and Cl2 are at Equilibrium at 500K in a closed container and their concentrations are 0.8 × 10-3 mol L-1, 1.2 × 10-3 mol L-1 and 1.2 × 10-3 mol L-1 respectively. The value of Kc for the reaction PCl5 (g) ⇋ PCl3 (g) + Cl2 (g) will be
view answer >Which of the following statements is incorrect?
view answer >When hydrochloric acid is added to cobalt nitrate solution at room temperature, the following reaction takes place and the reaction mixture becomes blue. On cooling the mixture it becomes pink. On the basis of this information mark the correct answer
[Co (H2O)6]3+ (aq) + 4Cl– (aq) ⇋ [CoCl4] 2- (aq) + 6H2O (l)
(pink) (blue)
view answer >
The pH of neutral water at 25°C is 7.0. As the temperature increases, ionisation of water increases, however, the concentration of H+ ions and OH– ions are equal. What will be the pH of pure water at 60°C?
view answer >The ionisation constant of an acid, Ka, is the measure of the strength of an acid. The Ka values of acetic acid, hypochlorous acid and formic acid are 1.74 × 10-5, 3.0 × 10-8 and 1.8 × 10-4 respectively. Which of the following orders of pH of 0.1 mol dm–3 solutions of these acids is correct?
view answer >Ka1, Ka2 and Ka3 are the respective ionisation constants for the following reactions
H2 S ⇋ H+ + HS-
HS ⇋ H+ + S2-
H2S ⇋ 2H+ + S2-
The correct relationship between Ka1 , Ka2 and Ka3 is
view answer >
The acidity of BF3 can be explained on the basis of which of the following concepts?
view answer >Which of the following will produce a buffer solution when mixed in equal volumes?
view answer >In which of the following solvents is silver chloride most soluble?
view answer >What will be the value of pH of 0.01 mol dm-3 CH3COOH (Ka = 1.74 × 10-5 )?
view answer >Ka for CH3COOH is 1.8 × 10-5 and Kb for NH4OH is 1.8 × 10-5. The pH of ammonium acetate will be
view answer >Which of the following options will be correct for the stage of half completion of the reaction A ⇋ B.
view answer >On increasing the pressure, in which direction will the gas-phase reaction proceed to re-establish Equilibrium, is predicted by applying Le Chatelier’s principle. Consider the reaction.
N2 (g) + 3H2 (g) ⇋ 2NH3 (g)
Which of the following is correct, if the total pressure at which the Equilibrium is established, is increased without changing the temperature?
view answer >
What will be the correct order of vapour pressure of water, acetone and ether at 30°C? Given that among these compounds, water has a maximum boiling point and ether has a minimum boiling point?
view answer >At 500 K, Equilibrium constant, Kc, for the following reaction is 5.
1/2 H2(g) + 1/2 I2 (g) ⇋ HI (g)
What would be the Equilibrium constant Kc for the reaction
2HI (g) ⇋ H2(g) + I2 (g)
view answer >
In which of the following reactions, the Equilibrium remains unaffected on the addition of a small amount of argon at constant volume?
view answer >Multiple Choice Questions II
For the reaction N2O4 (g) ⇋ 2NO2 (g), the value of K is 50 at 400 K and 1700 at 500 K. Which of the following options is correct?
view answer >At a particular temperature and atmospheric pressure, the solid and liquid phases of a pure substance can exist in Equilibrium. Which of the following term defines this temperature?
view answer >Short Answer
The ionization of hydrochloric in water is given below:
HCl (aq) + H2O (l) ⇋ H3 O+ (aq) + Cl- (aq)
Label two conjugate acid-base pairs in this ionization.
view answer >
The aqueous solution of sugar does not conduct electricity. However, when sodium chloride is added to water, it conducts electricity. How will you explain this statement on the basis of ionization and how is it affected by the concentration of sodium chloride?
view answer >BF3 does not have proton but still acts as an acid and reacts with NH3. Why is it so? What type of bond is formed between the two?
view answer >Ionization constant of a weak base MOH, is given by the expression
Kb = ![]()
Values of ionisation constant of some weak bases at a particular temperature are given below:
Base Di-methylamine Urea Pyridine Ammonia
Kb 5.4 × 10-4 1.3 × 10-14 1.77× 10-9 1.77 × 10-5
Arrange the bases in decreasing order of the extent of their ionisation at Equilibrium. Which of the above base is the strongest?
view answer >
The conjugate acid of a weak base is always stronger. What will be the decreasing order of basic strength of the following conjugate bases?
OH-, RO-, CH3COO-, Cl-
view answer >
Arrange the following in increasing order of pH
KNO3 (aq), CH3COONa (aq), NH4Cl (aq), C6 H5COONH4 (aq)
view answer >
The value of Kc for the reaction 2HI (g) ⇋ H2 (g) + I2 (g) is 1 × 10-4
At a given time, the composition of the reaction mixture is
[HI] =2 × 10-5 mol, [H2] =1 × 10-5 mol and [I2] =1 × 10-5 mol. In which direction will the reaction proceed?
view answer >
On the basis of the equation pH = – log [H+], the pH of 10-8 mol dm-3 solution of HCl should be 8. However, it is observed to be less than 7.0. Explain the reason.
view answer >The pH of a solution of a strong acid is 5.0. What will be the pH of the solution obtained after diluting the given solution a 100 times?
view answer >A sparingly soluble salt gets precipitated only when the product of the concentration of its ions in the solution (Qsp) becomes greater than its solubility product. If the solubility of BaSO4 in water is 8 × 10-4 mol dm-3, calculate its solubility in 0.01 mol dm-3 of H2SO4.
view answer >pH of 0.08 mol dm-3 HOCl solution is 2.85. Calculate its ionisation constant.
view answer >Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.
view answer >The solubility product of Al(OH) 3 is 2.7 × 10-11. Calculate its solubility in gL-1 and also find out the pH of this solution. (Atomic mass of Al = 27 u).
view answer >Calculate the volume of water required to dissolve 0.1 g lead (II) chloride to get a saturated solution. (Ksp of PbCl2 = 3.2 × 10-8, atomic mass of Pb = 207 u).
view answer >A reaction between ammonia and boron trifluoride is given below:
: NH3 + BF3→ H3N: BF3
Identify the acid and base in this reaction. Which theory explains it? What is the hybridisation of B and N in the reactants?
view answer >
Following data is given for the reaction: CaCO3 (s) → CaO3 (s) + CO2 (g)
ΔfH⊖ [CaO (s)] = -635.1 kJ mol=-1
ΔfH⊖ [CO2 (g)] = -393.5 kJ mol-1
ΔfH⊖ [CaCO3 (s)] = -1206.9 kJ mol-1
Predict the effect of temperature on the Equilibrium constant of the above reaction.
view answer >
Matching Type
Match the following equilibria with the corresponding condition.
For the reaction: N2 (g) + 3H2 (g) ⇋ 2NH3 (g)
Equilibrium constant Kc =
Some reactions are written below in Column I and their Equilibrium constants in terms of Kc are written in Column II. Match the following reactions with the corresponding Equilibrium constant
Column I (Reaction) Column II (Equilibrium constant)

view answer >
Match standard free energy of the reaction with the corresponding Equilibrium constant
Match the following species with the corresponding conjugate acid
Species Conjugate acid
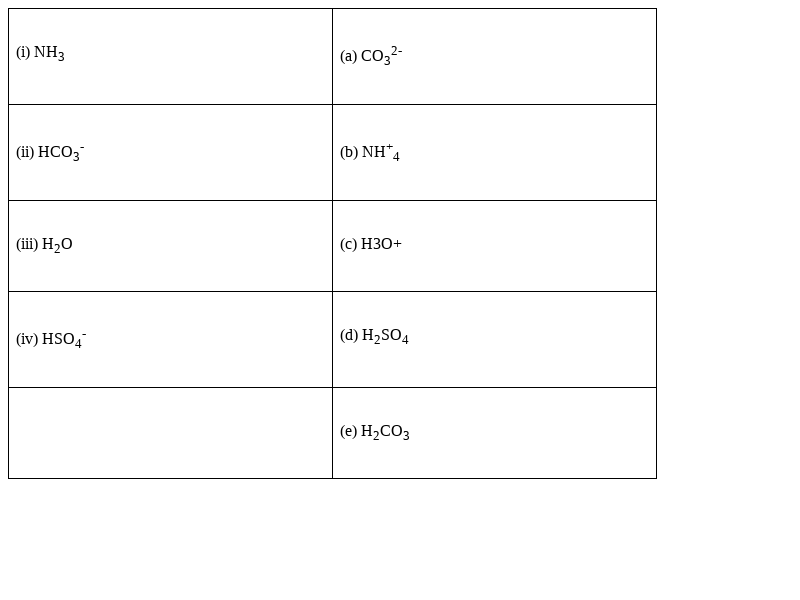
view answer >
Match the following graphical variation with their description
A B
(i)
 (a) Variation in product concentration with time
(a) Variation in product concentration with time
(ii)
 (b) Reaction at Equilibrium
(b) Reaction at Equilibrium
(iii)
 (c) Variation in reactant concentration with time
(c) Variation in reactant concentration with time
view answer >
Match Column (I) with Column (II)
Column I Column II
(i) Equilibrium (a) ∆G > 0, K < 1
(ii) Spontaneous reaction (b) ∆G = 0
(iii) Non spontaneous reaction (c) ∆G⊖ = 0
(d) ∆G < 0, K >1
view answer >
Assertion and Reason
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): Increasing order of acidity of hydrogen halides is HF < HCl < HBr < HI
Reason (R): While comparing acids formed by the elements belonging to the same group of periodic table, H–A bond strength is a more important factor in determining acidity of an acid than the polar nature of the bond.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A) : A solution containing a mixture of acetic acid and sodium acetate maintains a constant value of pH on addition of small amounts of acid or alkali.
Reason (R) : A solution containing a mixture of acetic acid and sodium acetate acts as a buffer solution around pH 4.75.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): The ionisation of hydrogen sulphide in water is low in the presence of hydrochloric acid.
Reason (R) : Hydrogen sulphide is a weak acid.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): For any chemical reaction at a particular temperature, the Equilibrium constant is fixed and is a characteristic property.
Reason (R) : Equilibrium constant is independent of temperature.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A) : Aqueous solution of ammonium carbonate is basic.
Reason (R) : Acidic/basic nature of a salt solution of a salt of weak acid and weak base depends on Ka and Kb value of the acid and the base forming it.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): An aqueous solution of ammonium acetate can act as a buffer.
Reason (R) : Acetic acid is a weak acid and NH4OH is a weak base.
view answer >
Note: In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Assertion (A): In the dissociation of PCl5 at constant pressure and temperature addition of helium at Equilibrium increases the dissociation of PCl5 .
Reason (R) : Helium removes Cl2 from the field of action.
view answer >
Long Answer
How can you predict the following stages of a reaction by comparing the value of Kc and Qc ?
(i) Net reaction proceeds in the forward direction.
(ii) Net reaction proceeds in the backward direction.
(iii) No net reaction occurs.
view answer >
On the basis of Le Chatelier principle explain how temperature and pressure can be adjusted to increase the yield of ammonia in the following reaction.
N2 (g) + 3H2 (g) ⇋ 2NH3 (g) Δ H = -92.38 kJ mil-1
What will be the effect of addition of argon to the above reaction mixture at constant volume?
view answer >
A sparingly soluble salt having general formula Ap+x Bq-y and molar solubility S is in Equilibrium with its saturated solution. Derive a relationship between the solubility and solubility product for such salt.
view answer >Write a relation between ∆G and Q and define the meaning of each term and answer the following :
(a) Why a reaction proceeds forward when Q < K and no net reaction occurs when Q = K.
(b) Explain the effect of increase in pressure in terms of reaction quotient Q. for the reaction: CO(g) + 3H2(g) + 3H2 (g) ⇋ CH4 (g) + H2 O (g)
view answer >