Potassium chlorate (KC103) on heating forms potassium chloride and oxygen. Write a balanced equation for this reaction and indicate the evolution of gas.
2KClO3 (s)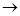 2KCl (s) + 302 (g)
2KCl (s) + 302 (g)
Here oxygen will be released in the form of O2 gas.
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.