Ammonia reacts with oxygen to form nitrogen and water. Write a balanced chemical equation for this reaction.
Add the state symbols for all the reactants and products.
4NH3 (g) + 302(g)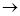 2N2 (g) + 6H20 (l)
2N2 (g) + 6H20 (l)
The equation is balanced and states of reactants and products are denoted.
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.