Write one equation each for the decomposition reactions where energy is supplied in the form of (a) heat, (b) light, and(c) electricity.
(a) Decomposition reaction, heat is used as energy.

(b) Light is supplied as energy
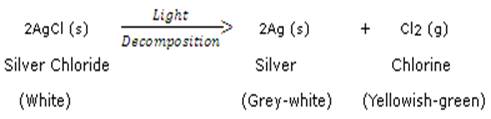
(c) Electricity is Supplied as energy
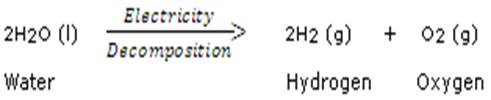
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.