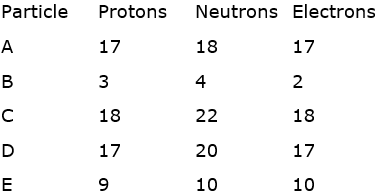
Composition of the nuclei of two atomic species A and B is given as under:

Give the mass numbers of A and B. What is the relation between the two species and which element or elements they represent?
The mass numbers of A and B is 40 the relation between the two species is that, they are isobars and the element or elements they represent are Argon and Calcium respectively.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.