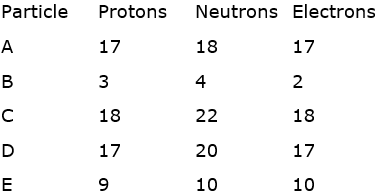
Which of the following pair is are isobars?
(i) ![]()
(ii) ![]()
Give reasons for your choice.
![]() , is a pair of isobar. The reason behind I, they both have same number of neutrons.
, is a pair of isobar. The reason behind I, they both have same number of neutrons.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.