The temperature and pressure at Shimla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Shimla.
We know ideal gas equation
PV=nRT
Where V= volume of gas
R=gas constant
T=temperature
n=number of moles of gas
P=pressure of gas
Number of moles n =![]() =
=![]()
Density ![]()
Given:
Temperature of Shimla T1=15.0°C
T(K)=T (![]() )+273.15
)+273.15
T1=T(K)=15+273.15=288.15K
Pressure of Shimla P1 = 72.0 cm of mercury
Temperature of Kalka T2 = 35.0°C
T(K)=T (![]() )+273.15
)+273.15
T2=T(K)=35+273.15=308.15K
Pressure of Kalka P2=76.0 cm of mercury
Substituting the value of n and ![]() in ideal gas equation, we get
in ideal gas equation, we get
![]() =
=![]()
So,
![]()
![]()
![]()
Taking the ratio of equations (i) and (ii),
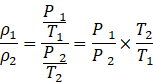
![]()
![]()
![]() The ratio of air density at Kalka to the air density at Shimla is 0.98.
The ratio of air density at Kalka to the air density at Shimla is 0.98.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
