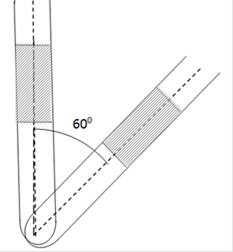
An ideal gas is trapped between a mercury column and the closed-end of a narrow vertical tube of uniform base containing the column. The upper end of the tube is open to the atmosphere. The atmospheric pressure equals 76 cm of mercury. The lengths of the mercury column and the trapped air column are 20 cm and 43 cm respectively. What will be the length of the air column when the tube is tilted slowly in a vertical plane through an angle of 60°? Assume the temperature to remains constant.
Let curved surface area of tube =A
Given
Length of air column=43cm=0.43m
Length of mercury column=20cm=0.20m
Pressure due to mercury column=![]() =0.2m of Hg
=0.2m of Hg
Atmospheric pressure=Pa=0.76m of Hg
Let the pressure of air column before titling =P1
So P1=Pa +PH
P1=0.76+0.2=0.96m of Hg
Volume =area![]() height
height
Volume of trapped air V1=A![]() length of air column=0.43A
length of air column=0.43A
If the tube is titled through an angle 60o only pressure of mercury column will get affected and not the atmospheric pressure.
So, change in PH will be
P’H=PHcos60o = 0.2![]() 0.5 =0.1m of Hg
0.5 =0.1m of Hg
So now the pressure of air column will become P2
P2=Pa+P’H=0.76+0.1=0.86m of Hg
Then volume will change. Let it now be V2=lA where l is new length of air column.
It is given in question that the temperature remains same. So, according to boyle’s law which states that PV=constant when temperature is constant, we can write,
P1V1=P2V2
![]()
![]()
![]()
![]() Length of the air column will become 0.48m=48cm
Length of the air column will become 0.48m=48cm
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.