(i) Graphically explain the effect of temperature on the rate constant of reaction? How can this temperature effect on rate constant be represented quantitatively?
ii) The decomposition of a hydrocarbon follows the equation
K = ( 4.5 X 1011 S -1) e-28000/KT
Calculate Ea.
OR
i) In the reaction
Q + R → Products
The time taken for 99% reaction of Q is twice the time taken for 90% reaction of Q.
The concentration of R varies with time as shown in the figure below:

What is the overall order of the reaction? Give the units of the rate constant for the same. Write the rate expression for the above reaction.
ii) Rate constant for a first order reaction has been found to be 2.54 x 10-3s-1. Calculate its three- fourth life.
(i)
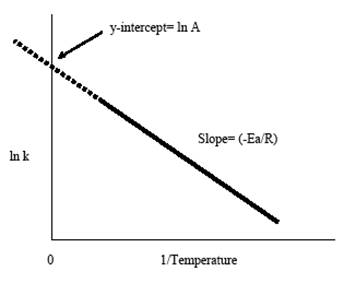
• Rate constant of a reaction depends on temperature and it is said that rate constant increases almost doubly for evey increase of 10 ◦ of temperatureand this dependency can be expressed quantitatively by Arrhenius equation :
k = A e – Ea/RT
Where , K= rate constant, A= frequency factor or Arrhenius factor R= universal gas constant and T= temperature (Kelvin) , Ea = energy of activation for the reaction.
• To represent this relationship graphically we have to reform the equation as :
By taking ln on the both sides we get,
lnK = - Ea/RT + lnA
and by plotting ln k values of a certain reaction against the (1/T) values we get the following straight line graph with a negative slope = - Ea/R and an intercept of ln A
 .
.
(ii)
• From Arrhenius equation k = A e – Ea/RT .
• R= N0K , where R = ideal gas constant , k= boltzman constant and N0 = avogadro’s number.
Hence K =R/ N0 .
From the given equation , e – Ea/kT = e-28000N0/RT = e – Ea/RT
Hence ,
![]() =
= ![]()
Or, Ea = 28000 X 6.023 X 1023
=1.744 X 1028 joule/molecule .
OR
(i)
• From the given plot the order of the reaction should be of zero order.
Hence, Q + R → Products is a zero order reaction .
• Because, the concentration of R ,
[R] varies with time which is shown by the plot. And that means the rate of the reaction is actually proportional to the concentration of reactant R (zero power on [R] ).
i.e. rate = d[R]/dt =k R0 = K (R0 = 1] which upon integration results in,
[R] = -kt + [R]0( where , [R] is the final concentration and [R]0 is the initial concentration
or, k = [R] – [R]0
t
• The dimension of the rate constant for the reaction is concentration/time hence, unit = M/s.
• The rate expression for the given zero order reaction is ;
rate = d[R]/dt =kR0 = K
hence, rate=k
(ii)
• For the given 1st order reaction K=2.54 x 10-3 s-1
• For a 1st order reaction t= 2.303 log a
k a-x
where, a = initial amount of the reactant and a-x denotes final amount until the reaction is taken into consideration.
• Now , for 3/4 th life a – x =a – (3/4) a= a/4
• t3/4 = 2.303 log a
2.54x10-3 a/4
= 2.303 log 4
2.54x10-3
=2.303 X 0.6020 [ log 4 =0.6020]
2.54x10-3
= 545.82 second.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.





