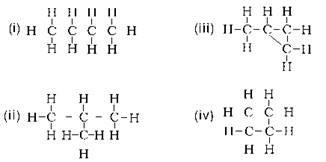
Which of the following statements are usually correct for carbon compounds? These
(i) are good conductors of electricity
(ii) are poor conductors of electricity
(iii) have strong forces of attraction between their molecules
(iv) do not have strong forces of attraction between their molecules
Carbon compounds are poor conductors of electricity because they form covalent bonds. In such bonds, there are no free electrons left as all are used in making the bond. Hence carbon compounds do not dissociate into ions and hence are poor conductors of electricity. Moreover, covalent bonds do not have strong forces of attraction. A covalent bond is also known as a molecular bond.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.