State briefly how you will extract iron from its ore. Draw a neat and labelled diagram. Write all the chemical reactions involved in it.
Iron is usually extracted from its chief ore, Haemitite.
The various steps involved in the production of iron metal from haematite are as follows: 1. Concentration of ore
The concentration of haematite or is done by the method of hydraulic washing. The ore is spilt into small pieces and then washed in a stream of water to remove sand, clay. In this way , a fairly concentrated ore is obtained and usually there is no need of any further concentration.
2. Calcination
The washed iron ore is then strongly heated in the absence of air to expel water sticking to it.
3. Reduction
The washed and dried ore is mixed with weighed quantities of coke and limestone and put into a blast furnace from the top .A blast of hot air is blown into the furnace from near its bottom. This air is to supply for the burning of coke.
The two reactions which take place in the blast furnace leading to the formation of iron metal are: formation of carbon monoxide and reduction of haematite.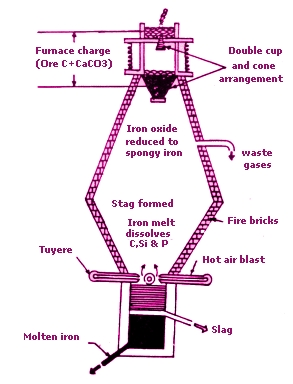 1) Formation of carbon monoxide
1) Formation of carbon monoxide
Carbon monoxide is the main reducing agent in the extraction of iron by blast furnace. Carbon monoxide is formed from coke in two steps:
Near the bottom of the furnace coke burns in air to form carbon dioxide and a lot of heat is produced
C (s) + O2 CO2 + Heat
CO2 + Heat
In the center of the furnace, carbon dioxide reacts with white hot coke to form carbon monoxide.
CO2 + C(s) 2 CO2) Reduction of Iron (III) oxide to Iron
2 CO2) Reduction of Iron (III) oxide to Iron
In the upper part of the furnace, carbon monoxide reduces iron (III) oxide to iron metal
Fe2O3 + 3CO 2Fe(l) +3CO2(g)
2Fe(l) +3CO2(g)
Iron metal is produced as a grey solid which melts as it comes down in the furnace where the temperature is very high. Molten iron collects at the bottom of the furnace.
The various steps involved in the production of iron metal from haematite are as follows: 1. Concentration of ore
The concentration of haematite or is done by the method of hydraulic washing. The ore is spilt into small pieces and then washed in a stream of water to remove sand, clay. In this way , a fairly concentrated ore is obtained and usually there is no need of any further concentration.
2. Calcination
The washed iron ore is then strongly heated in the absence of air to expel water sticking to it.
3. Reduction
The washed and dried ore is mixed with weighed quantities of coke and limestone and put into a blast furnace from the top .A blast of hot air is blown into the furnace from near its bottom. This air is to supply for the burning of coke.
The two reactions which take place in the blast furnace leading to the formation of iron metal are: formation of carbon monoxide and reduction of haematite.
 1) Formation of carbon monoxide
1) Formation of carbon monoxideCarbon monoxide is the main reducing agent in the extraction of iron by blast furnace. Carbon monoxide is formed from coke in two steps:
Near the bottom of the furnace coke burns in air to form carbon dioxide and a lot of heat is produced
C (s) + O2
 CO2 + Heat
CO2 + HeatIn the center of the furnace, carbon dioxide reacts with white hot coke to form carbon monoxide.
CO2 + C(s)
 2 CO2) Reduction of Iron (III) oxide to Iron
2 CO2) Reduction of Iron (III) oxide to IronIn the upper part of the furnace, carbon monoxide reduces iron (III) oxide to iron metal
Fe2O3 + 3CO
 2Fe(l) +3CO2(g)
2Fe(l) +3CO2(g)Iron metal is produced as a grey solid which melts as it comes down in the furnace where the temperature is very high. Molten iron collects at the bottom of the furnace.
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.