What do you mean by covalent bonding?
The bonding in which atoms mutually share the electron pair(s) to form molecules is called covalent bonding.
Example: Hydrogen molecule. In hydrogen molecule, both the electrons are shared to form a covalent bond.

Another very common example is the water molecule:
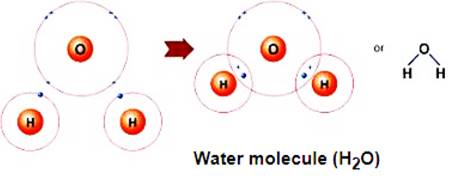
In water molecule, the two single hydrogen & oxygen bonds are formed.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.