Draw the structure of diamond.
In diamond, each carbon atom is covalently bonded to four other carbon atoms.
The crystal structure is shown below:
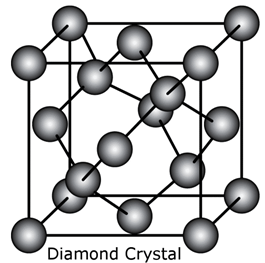
Diamond has a 3D structure.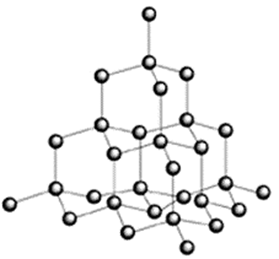
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.