Write the two examples of the crystalline form of carbon.
In the crystalline form of carbon, many atoms are bonded together in a repeating pattern. The arrangement of atoms in the crystal differs for each form of carbon. Two forms are:
i. Diamond.
Each carbon atom is covalently bonded to four other carbon atoms. The arrangement of carbon is shown below:
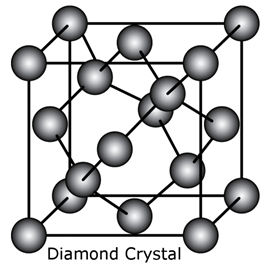
ii. Fullerene:
each carbon atom is covalently bonded to four other carbon atoms. Each sphere contains 60 carbon atoms, and each carbon atom is bonded to three others by single covalent bonds.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.