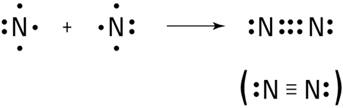
Write the covalent bonding in nitrogen.
Nitrogen has the electronic configuration 2, 5. It shares 3 of the outermost electrons with another nitrogen atom to form a triple-bonded N2 molecule, as shown below:
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.