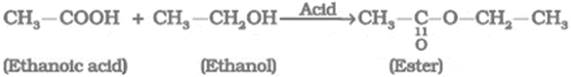
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
When ethanol reacts with ethanoic acid in the presence of concentrated sulphuric acid (a catalyst), a sweet smelling ester (CH3CH2COOCH3) is produced.
AI is thinking…
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.