Figure shows an adiabatic cylindrical tube of volume V0 divided in two parts by a frictionless adiabatic separator. Initially, the separator is kept in the middle, an ideal gas at pressure p1 and temperature T1 is injected into the left part and another ideal gas at pressure p2 and temperature T2 is injected into the right part. CP/CV = γ is the same for both the gases. The separator is slid slowly and is released at a position where it can stay in equilibrium. Find
(a) the volumes of the two parts,
(b) the heat given to the gas in the left part
(c) the final common pressure of the gases.
![]()
Given:
An adiabatic cylindrical tube of volume V0 is divided in two parts by a frictionless adiabatic separator.
An ideal gas at pressure p1 and temperature T1 is injected into the left part and another ideal gas at pressure p2 and temperature T2 is injected into the right part.
(a) When the piston is slowly moved to the equilibrium position, one side increases in volume when the other side decreases.
The processes will be adiabatic,
For the left part,
![]() Where, subscript 1 and 2 represent the initial and the final state.
Where, subscript 1 and 2 represent the initial and the final state.
⇒ ![]() ……….(1)
……….(1)
And for the right part,
![]() ……….(2)
……….(2)
We are assuming P to be the common pressure.
Dividing (1) by (2) we get,
![]()
Again,
![]()
So,
⇒ ![]()
⇒ 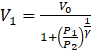
Therefore, 
The final volume of the left and the right side will be 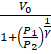 and
and  respectively.
respectively.
(b) The heat given will be zero as the whole process is taking place in an adiabatic surrounding.
(c) So putting the above result in (1) we get,
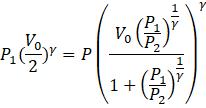
⇒ 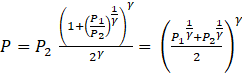
Thus the final common pressure of the gasses will be 
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
