An adiabatic cylindrical tube of cross-sectional area 1 cm2 is closed at one end and fitted with a piston at the other end. The tube contains 0.03g of an ideal gas. At 1 atm pressure and at the temperature of the surrounding, the length of the gas column is 40 cm. The piston is suddenly pulled out to double the length of the column. The pressure f the gas falls to 0.355 atm. Find the speed of sound in the gas at atmospheric temperature.
Given:
An adiabatic cylindrical tube of cross-sectional area 1 cm2 is closed at one end and fitted with a piston at the other end.
The tube contains 0.03g of an ideal gas at 1 atm pressure and at the temperature of the surrounding.
The length of the gas column is 40 cm.
The piston is suddenly pulled out to double the length of the column and the pressure of the gas falls to 0.355 atm.
The expansion process of the gas is adiabatic, so ![]() Where, subscript 1 and 2 represent the initial and the final state.
Where, subscript 1 and 2 represent the initial and the final state.
When the length is increased to double the volume is also doubled.
So,
![]()
⇒ ![]()
⇒ ![]()
⇒ ![]()
The speed of sound in gas at atmospheric pressure is given as
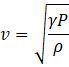
where γ is the adiabatic constant, P is pressure and ρ is the volume density. The speed is
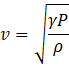
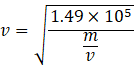
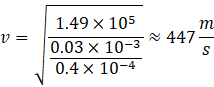
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.