Arrange the following:
(i) in increasing order of basic strength
![]()
(ii) in increasing order of boiling point
![]()
Basicity of any compound depends on the following factors:
(i) the ease with which it can donate electrons
(ii) the impact inductive effect or resonance effect
(iii) electronegativity of the atom
From the given compound aniline is aromatic, the lone pair over nitrogen is in resonance with the benzene ring which reduces its tendency for donating that electrons.
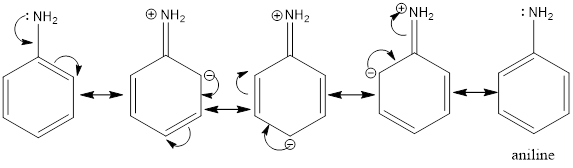
While the methyl-phenyl amine or N-methylaniline also has its lone pair in resonance the positive charge on nitrogen is balanced by the positive inductive effect from methyl group. So, it is a stronger base than aniline.

And among the three compounds, ethylamine is the strongest base as the electron density is increased by the methyl group by +I effect.
Thus the order of basicity is
![]()
Option (ii)
Boiling point is dependent on the molar mass of the compound and its structure. Higher molar mass generally implies higher boiling point, but for the case of same or similar molar mass we consider branching and the nature of the compounds and the interatomic and intermolecular attraction.
In case of alcohol they undergo intramolecular hydrogen bonding which increases their boiling point significantly. In fact out of hydrides of group 16, water has shown abnormality and has the highest boiling point in the group.
So among the three compounds the highest boiling point is of ethanol. The branching in N,N-dimethyl amine decreases its boiling point as branching leads to more occupancy per molecule which decreases its density. Thus the order for boiling point is:
![]()
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.