Explain the difference between Buna – N, and Buna – S.
Buna-N:
The monomers are as follows:
Butadiene- CH2 = CH-CH = CHs
Acrylonitrile- CH2 = CH - CN
The reaction is as follows:
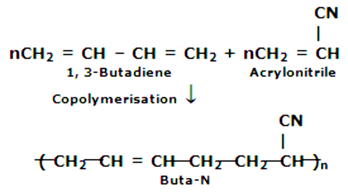
This polymer is also called as nitrile rubber and this used in the manufacture of oil seals, protective gloves etc.
Buna – S:
The monomers are as follows:
1, 3- Butadiene
Styrene
The reaction as follows:
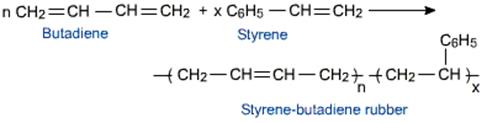
It is also called as Styrene Butadiene Rubber and it possess less tensile strength in comparison with natural rubber and it is used in the manufacture of automobile tyres etc.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.