The formula of carbonate of a metal M is M2CO3. What is the formula for phosphate of M?
It is given that carbonate of a metal M is M2CO3.
By applying criss cross method, we can know the valency of “M”:

Hence, the valency of “M” is +1
Now, formula for phosphate of metal M:
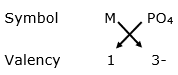
Formula: M3PO4
Thus, the formula for phophate of metal is M3PO4.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.