An element X has a valency 1.
(i) Write the chemical formula of its phosphide.
(ii) Write the chemical formula of its chloride.
(iii) Is element X a metal or a non-metal?
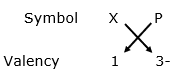
Chemical formula: X3P
(ii) 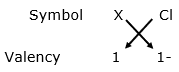
Chemical formula: XCl
(iii) The element X is metal.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.