In a chemical compound calcium sulphate:
(i) Identify the two ions.
(ii) Write the chemical formula of compound formed when positive ion is replaced by sodium ion.
(iii) Name the resulting compound.
(i) The two ions are Ca2+ (cation) and SO42-(anion)
(ii) When positive ion is replaced by sodium ion:
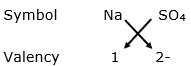
Chemical formula: Na2SO4
(iii) The name of the resulting compound (Na2SO4) is sodium sulphate.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.