If an element X has a valency of 3, then find out the formula of its chloride and sulphide.
It is given that element X has a valency of 3.
Formula for chloride of element X:

Formula: MCl3
Thus, the formula for chloride of element X is MCl3.
Formula for sulphide of element X:
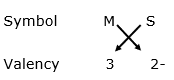
Formula: M2S3
Thus, the formula for sulphide of element X is M2S3.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
