Write the chemical formulae and names of the compounds formed by the following ions:
(i) Cr3+ and SO42–
(ii) Pb2+ and NO3–
(iii) Mg2+ and CO32–
(i) Cr3+ and SO42-
By applying criss-cross method:
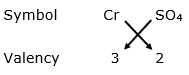
Chemical formula: Cr2(SO4)3
Name of the compound: Chromium sulphate
(ii) Pb2+ and NO3–
By applying criss-cross method:

Chemical formula: Pb(NO3)
Name of the compound: Lead nitrate
(iii) Mg2+ and CO32–
By applying criss-cross method:

Chemical formula: MgCO3
Name of the compound: Magnesium carbonate
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.