Write the chemical formulae of Nitrates (NO3–) of Na+, K+, Al3+, Mg2+, Ca2+, Zn2+.
(i) For Na+
By applying criss-cross method:
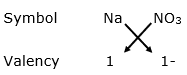
Formula: NaNO3
(ii) For K+
By applying criss-cross method:
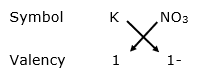
Formula: KNO3
(iii) For Al3+
By applying criss-cross method:
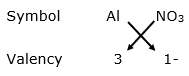
Formula: Al(NO3)3
(iv) For Mg2+
By applying criss-cross method:

Formula: Mg(NO3)2
(v) For Ca2+
By applying criss-cross method:
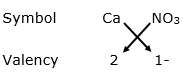
Formula: Ca(NO3)2
(vi) For Zn2+
By applying criss-cross method:

Formula: Zn(NO3)2
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.