Write the structure of Monomers used for getting the following polymers:
(a) Novolac
(b) Neoprene
(c) Buna - S
OR
(a) Write one example each of
(i) Cross-linked polymer.
(ii) Natural Polymer
(b) Arrange the following in the increasing order of their intermolecular forces:
Terylene, Buna - N, Polystyrene
(c) Define biodegradable polymers with an example.
(a) The monomers of novolac are o-and/or p-hydroxymethylphenol derivatives.
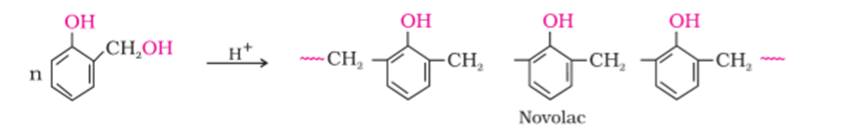
(b) The monomer of neoprene is 2- Chloro- 1, 3- butadiene.
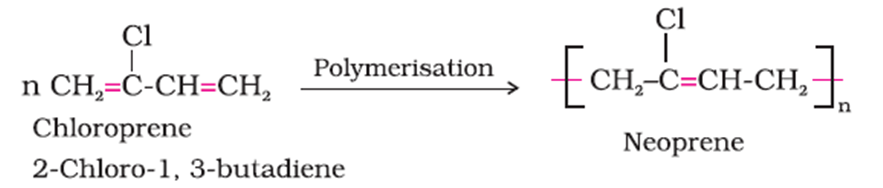
(c) The monomers of Buna- S are styrene and 1,3- Butadiene.
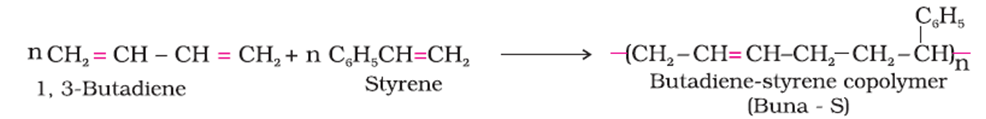
OR
(a) (i) Bakelite
(ii) Proteins
(b) Buna-N s a type of rubber, Polystyrene is a type of thermoplastic polymer and Terylene is a type of fibre.
The intermolecular forces are strongest in fibres then in plastics and weakest in rubbers. Thus, polymers in increasing order of intermolecular forces are arranged as follows-
Buna-N < Polystyrene < Terylene
(c) Biodegradable polymers are those polymers which can be decomposed by microorganisms with time and do not cause pollution. E.g. PHBV, Nylon 2–nylon 6, etc
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.
