With the help of an activity show the blue colour of the sky and the reddish appearance of the sun at the sunrise or sunset.
i. The molecules of air and other fine particles in the atmosphere have a size smaller than the wavelength of visible light. These are more effective in scattering light of shorter wavelengths at the blue end than the light of longer wavelengths at the red end.
ii. The red light has a wavelength about twice that of blue light. Thus, when sunlight passes through the atmosphere, the fine particles in the air scatter the blue color (shorter wavelengths) more strongly than red.
iii. The scattered blue light enters our eyes. Light from the Sun near the horizon passes through thicker layers of air and larger distance in the earth’s atmosphere before reaching our eyes. However, light from the Sun overhead would travel a relatively shorter distance.
iv. At noon, the Sun appears white as only a little of the blue and violet colors are scattered. Near the horizon, most of the blue light and shorter wavelengths are scattered away by the particles. Therefore, the light that reaches our eyes is of longer wavelengths. This gives rise to the reddish appearance of the Sun.
The following activity illustrate the above effect: -
1. Place a strong source (S) of white light at the focus of a converging lens (L1). This lens provides a parallel beam of light.
2. 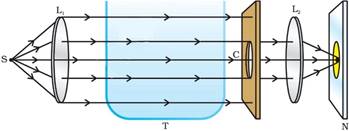
3. Allow the light beam to pass through a transparent glass tank (T) containing clear water.
4. Allow the beam of light to pass through a circular hole (c) made in a cardboard. Obtain a sharp image of the circular hole on a screen (MN) using a second converging lens (L2), as shown in the diagram.
5. Dissolve about 200 g of sodium thiosulphate (hypo) in about 2 L of clean water taken in the tank. Add about 1 to 2 mL of concentrated Sulphuric acid to the water.
Observations: -
It is observed that the microscopic Sulphur particles start precipitating in about 2 to 3 minutes. As the Sulphur particles begin to form, it is observed that the blue light from the three sides of the glass tank. This is due to scattering of short wavelengths by minute colloidal Sulphur particles. The color of the transmitted light as observed from the fourth side of the glass tank facing the circular hole at first is orange red color and then slowly changes to bright crimson red color on the screen.
Couldn't generate an explanation.
Generated by AI. May contain inaccuracies — always verify with your textbook.